 Write the electron configuration for the. Mårtensson, "Core-Level Binding Energies in Metals," J. ScienceChemistryWrite the electron configuration for the ground state of Co (cobalt 4+). The concept of electronic configuration has replaced the older concept of valency and valence electrons. Lide, (Ed.) in Chemical Rubber Company handbook of chemistry and physics, CRC Press, Boca Raton, Florida, USA, 81st edition, 2000. The electronic configuration of each element is decided by the Aufbau principle which states that the electrons fill orbitals in order of increasing energy levels. 
Ley, Eds., Photoemission in Solids I: General Principles (Springer-Verlag, Berlin) with additional corrections, 1978. Burr, "Reevaluation of X-Ray Atomic Energy Levels," Rev. In this case of Cobalt there are 27 electrons which are present in 4 orbits and their distribution on the orbit that is electronic configuration can be written as: 1s 2 2s 2 2p 6 3s 2 3p 6 3d 7 4s 2. They are tabulated elsewhere on the WWW (reference 4) and in paper form (reference 5). The data are adapted from references 1-3. I am grateful to Gwyn Williams (Jefferson Laboratory, Virginia, USA) who provided the electron binding energy data. The binding energies are quoted relative to the vacuum level for rare gases and H 2, N 2, O 2, F 2, and Cl 2 molecules relative to the Fermi level for metals and relative to the top of the valence band for semiconductors. Then, the next electron leaves the 3d orbital and the configuration becomes: Ar4s 0 3d 6. Shorthand configuration Full configuration: 1s 2 2s 2 2p 6 3s 2 3p 6 3d 7 4s 2: Electron configuration The electronic configuration of cobalt is: 1s2 2s2. The first two to go are from the 4s orbital and Cobalt becomes:Ar4s 0 3d 7. When observing Cobalt 3+, we know that Cobalt must lose three electrons. All values of electron binding energies are given in eV. The electron configuration of Cobalt is Ar4s 2 3d 7. 1967, 47, 1300.Įlectron binding energies Electron binding energies for cobalt. its a d-block element.so electron is get added in 3d sub level energy.but 4s orbital is filled first, then 3d orbital. You have signed an examinee agreement, and it will be enforced on this subreddit.ĭo not intentionally advertise paid or free products or services of any sort.These effective nuclear charges, Z eff, are adapted from the following references: Answer (1 of 14): As cobalt (Co) has atomic number 27. We have one "stickied" post for each exam and score release day, contain all test day discussion/reactions to that thread only.ĭo not discuss any specific information from your actual MCAT exam. Ground state electron configurations are the foundation for understanding molecular bonding, properties, and structures. For an example format for submitting pictures of questions from practice material click hereĭo not link to content that infringes on copyright laws (MCAT torrents, third party resources, etc).ĭo not post repeat "GOOD LUCK", "TEST SCORE", or test reaction posts. These are considered spoilers and should be marked as such. Chemical Properties of Cobalt Electrochemical Equivalent: 1.0994g/amp-hr Electron Work Function: 5eV Electronegativity: 1.88 (Pauling) 1.7 (Allrod Rochow). Be nice to each other, hating on other users won't help you get extra points on the MCAT, so why do it?ĭo not post any question information from any resource in the title of your post. Rudeness or trolling will not be tolerated. Therefore, the valence electrons of cobalt are nine. Please message the moderators with your skills/ideas! MCAT RESOURCES & INFO Study Groups This electron configuration shows that the last shell of cobalt has two electrons and the d-orbital has a total of seven electrons. Want to help us improve this subreddit or tell us about a new resource we can add to the sidebar? 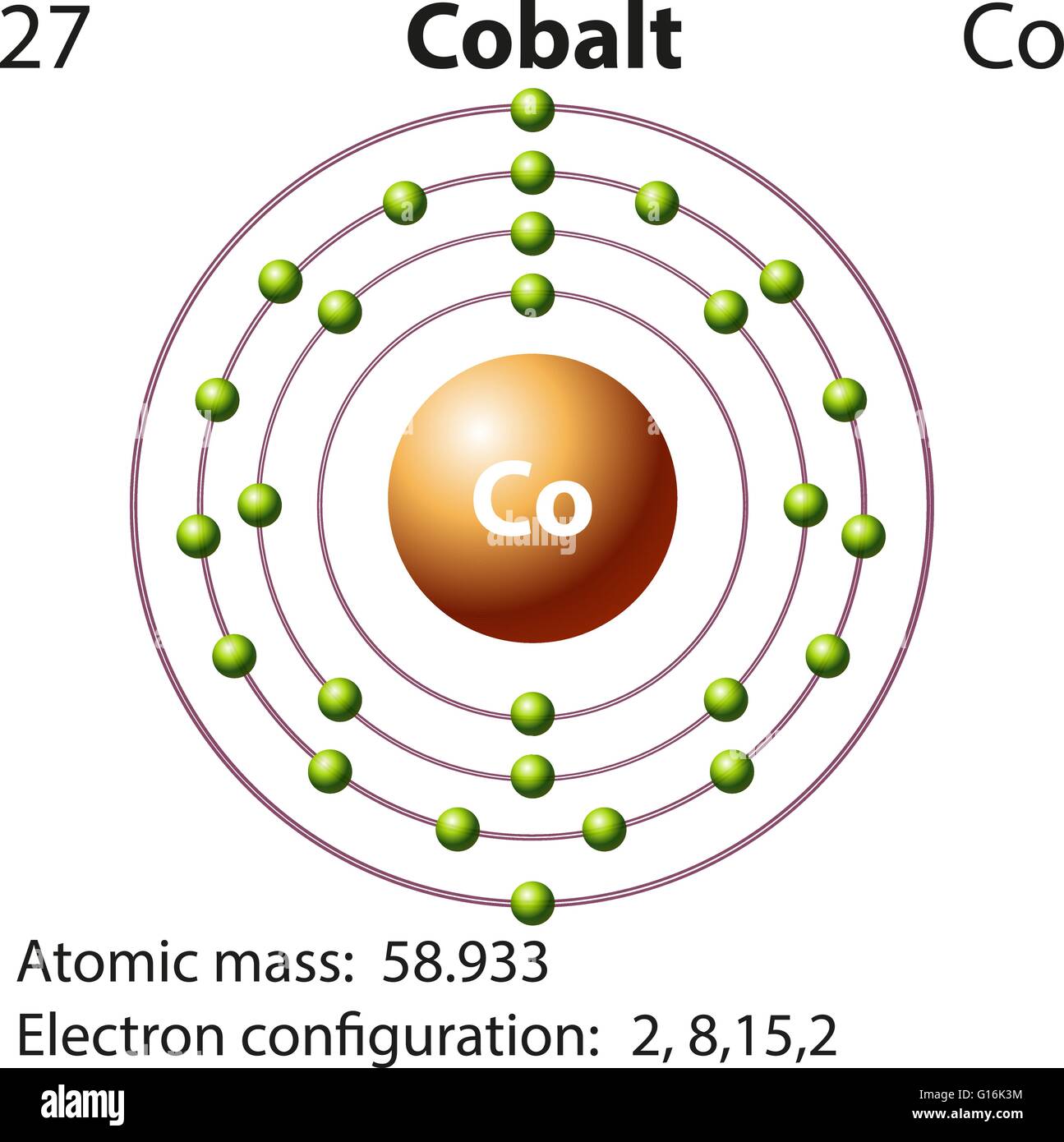 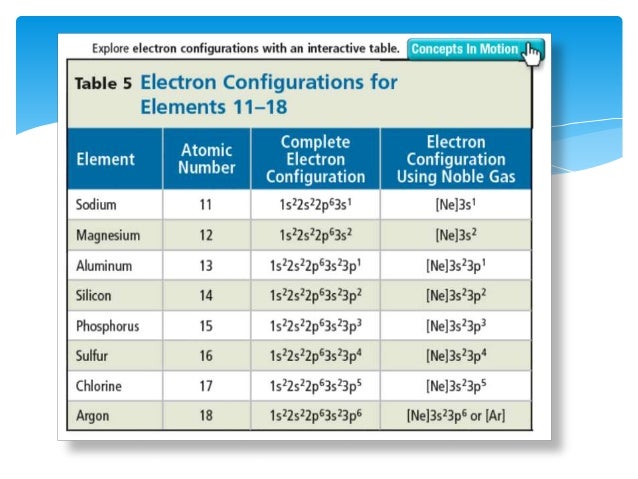
Below you will find our forum rules, resources, and more. We request that you read the sidebar COMPLETELY before you post. r/MCAT is a place for support, discussion, advice, social networking, news, study tips and more. The MCAT (Medical College Admission Test) is offered by the AAMC and is a required exam for admission to medical schools in the USA and Canada. When Nickel becomes Ni+2 Nickel has lost 2 electrons leaving the atom with only 8 valance electrons. 2 electrons in the 4s and 8 elections in the 3d. Welcome to the BEST place for MCAT prep and practice materials. Co (Cobalt) is an element with position number 27 in the periodic table. 18 electrons fill up the third electron shell leaving 10 valance electrons. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
